Study and Analysis of a Root Cause of Failure
in Resin Composite Materials
Galvanic Corrosion Resistance of Carbon Fiber Reinforced Plastics (CFRP)
When CFRP and a metal are joined, and then exposed to an electrolyte, galvanic corrosion can occur. JFE-TEC can study this type of corrosion and offer the remediation method.
Importance of evaluation of the galvanic corrosion resistance in CFRP
In recent years the mold insert molding process of which an insert material such as metal screw and ceramic is placed in a die in a mold followed by injection of plastic is commonly used and molded parts with insert are widely used in aircrafts. Use of CFRP in the molded parts with insert is increased in recent years in automobile industry.
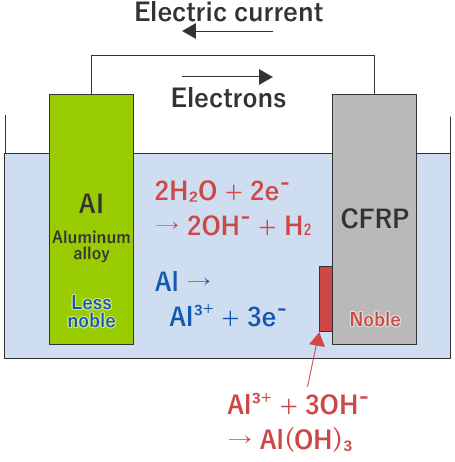
Carbon fibers are conductive with electric current and high in electric potential. In a CFRP molded part with a metal with the electric potential lower than carbon fibers wetting with rain or dew potentially causes the galvanic reaction to corrode (galvanic corrosion) the metal with a lower electric potential.
Countermeasures are required in the aircraft in which CFRP products are extensively used and similar measures will be required in the automobile parts in near future since more use of them are anticipated.
This clearly indicates the importance of evaluating the galvanic corrosion resistance of CFRP.
Evaluation of galvanic corrosion resistance
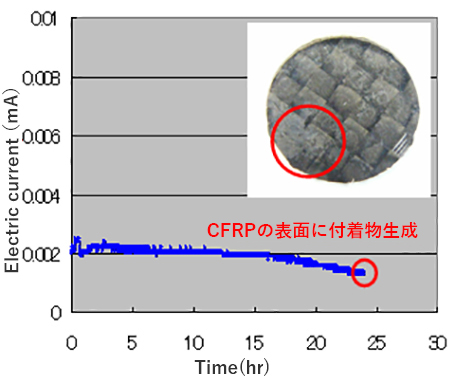
When two types of materials with different electric potentials and electric conductivity are joined in an aqueous electrolyte, the minute electric current (galvanic current) occurs. The electric potential generated depends on the length and the content of carbon fiber in CFRP and the galvanic current depends on the types of metals.
As illustrated in the following figure measurement of the electric potential and the galvanic current can determine the galvanic corrosion resistance of the CRFP with metals.

Measurement Example of Galvanic Current between CFRP and Aluminum Alloy
- (1) Condition:
- Aqueous 5% NaCl solution under atmosphere
- (2) Test sample:
- (1) Cathode: CFRP sheet, thickness = 1.0 mm, Prepreg matrix: Epoxyresin
(2) Anode: Aluminum alloy plate, thickness = 0.8 mm, Aluminum 5052
-

Scanning electron microscopy (SEM)-energy dispersive X-ray
spectroscopy (EDX) of the deposit on the CFRP surface -
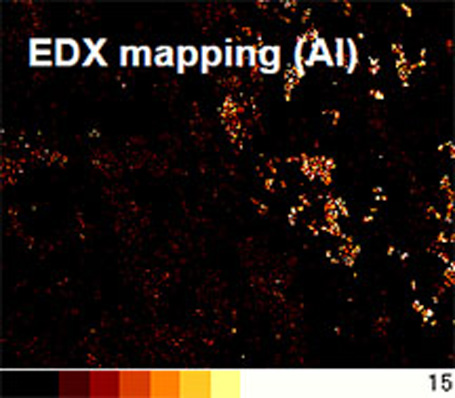
SEM-EDXS of the deposit on the CFRP surface